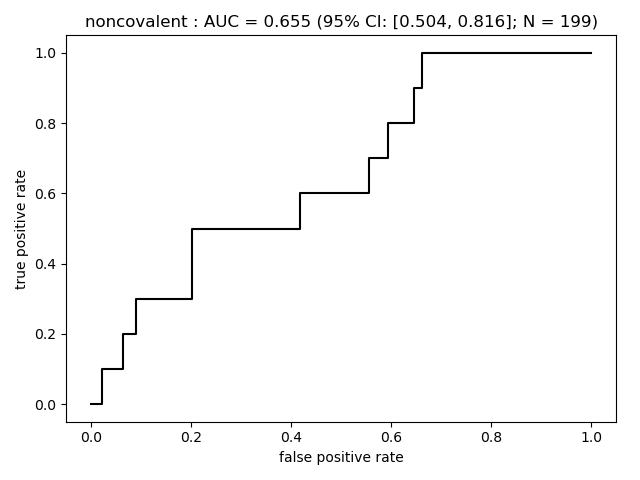
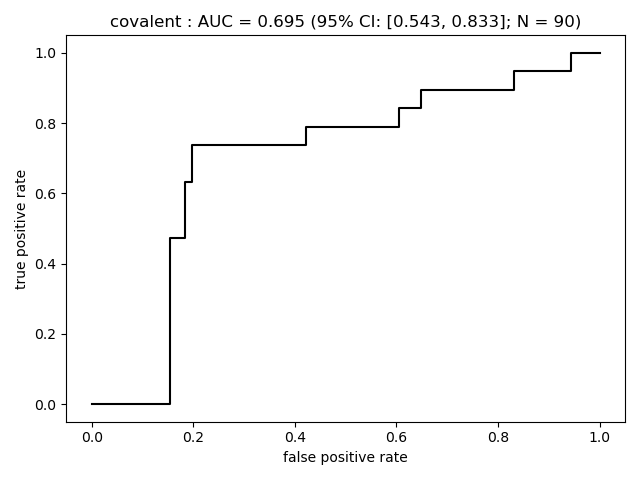
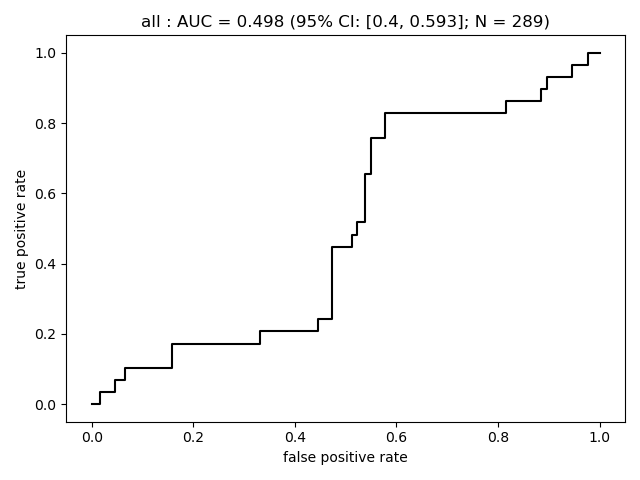
Summary
I used an ensemble hybrid docking protocol (built with the OpenEye Toolkit 2019.10.2) to dock the 2020-05-03 snapshot of COVID Moonshot compounds.
The CYS145(-) HIS41(+) form of Mpro monomer was used for docking.
All fragment structures were used hybrid docking (fragment shape/color overlay followed by docking minimization) to all fragment structures successfully prepared by SpruceTK using multiple likely protonation and tautomeric states, and the best-scoring pose was selected.
For covalent inhibitors, a constraint was imposed to place the relevant heavy atom for any covalent warhead within 4A of the CYS145 SG atom, rejecting poses that could not satisfy this constraint.
These docked poses were then solvated and simulated for 10 ns, and the minimum and average distances of the closest warhead heavy atom and CYS145 SG were reported.
Scores
The Chemgauss4 and CYS145-warhead dist mean (A) should be uploaded to CDD.
-
Chemgauss4is the Chemgauss4 docking score of the best scoring pose and protonation/tautomeric state to any Mpro structure; more negative is better, and fragments score ~ -10 -
docked_fragmentis the name of the fragment (and corresponding Mpro fragment structure) with the best scoring hybrid docking pose -
CYS145-warhead dist minimum (A)is the minimum distance (in Angstroms) between CYS145 SG and any covalent warhead heavy atom during 10 ns simulation, where distances < 4A indicate the warhead is potentially well-positioned for irreversible binding (covalent inhibitors only) -
CYS145-warhead dist mean (A)is the mean distance (in Angstroms) between CYS145 SG and any covalent warhead heavy atom during 10 ns simulation, where distances < 4A indicate the warhead is potentially well-positioned for irreversible binding (covalent inhibitors only) -
CYS145-warhead dist stddev (A)is the standard deviation (in Angstroms) of the distance between CYS145 SG and any covalent warhead heavy atom during 10 ns simulation, where large values may indicate the warhead samples a variety of distances from CYS145 (covalent inhibitors only)
Source and files
Edit: I’m unable to upload this file to fragalysis due to warnings of fragments and PDB structures that are unknown, but it can be downloaded here.
Edit: Covalent warhead distances have now been included.
All docking results will be posted here:
All scripts and receptor models are here:
Approach
Each fragment-bound X-ray structure provided by DiamondMX was prepared using the OpenEye Spruce Toolkit to clean up the structure and extract protein and small molecule structures with missing atoms added and missing loops either reconstructed or truncated.
CYS145 was deprotonated and the hydrogens reoptimized, which produced a doubly-protonated HIS41.
For each COVID Moonshot designed molecule, a set of reasonable protonation and tautomeric states were enumerated using OEGetReasonableProtomers from the OpenEye QuacPac Toolkit.
Each protomer was then expanded into a dense set of conformers using the OpenEye Omega Toolkit.
For each active site (non-covalent or covalent) and dimer interface fragment structure, one docked pose was generated using the binding site defined by the corresponding fragment using the corresponding X-ray structure.
Each multiconformer molecule was docked to the specified fragment site using the OpenEye OEDocking Toolkit using the Hybrid2 search method (which exploits shape overlays with the corresponding bound fragment) and the High search resolution.
The highest dock score across all docked structures was recorded, and the corresponding fragment structure noted.